Composition
- Each Film Coated Tablet Contains:
- Atorvastatin Calcium IP
50 mg
- Ezetimibe IP
10 mg
Packing
- 10x10
(Alu-Alu)
MRP
- 95
Overview
Before knowing its combination, we should need to know how they work individually so we know that Atorvastatin is a selective, competitive HMG-CoA reductase inhibitor. Atorvastatin is the only drug in its class specifically indicated for lowering both elevated LDL-cholesterol and triglycerides in patients with hypercholesterolemia. It prevents coronary atherosclerosis and reduces the risk of heart attack. Ezetimibe is in a class of lipid-lowering compounds that selectively inhibits the intestinal absorption of cholesterol and related phytosterols.
Indications?
• It is used for the treatment of dyslipidemia and the prevention of cardiovascular disease.• Ezetimibe is a prescription drug that is used to treat high cholesterol and a rare genetic condition called sitosterolemia.
Warnings
Sudden withdrawal of beta-adrnoceptor blocking agent in patients with ischaemic heart disease may result in the appearance of anginal attacks of increased frequency or severity or deteriorate in cardiac state. Discontinuation of therapy should be gradual.
Anaesthesia: Care should be taken when using anaesthetic agents with Atenolol. The anaesthetist should be informed to enable the necessary precautions to be taken.
Atenolol should only be used with caution in patients with controlled congestive cardiac failure or with a family history of asthma. Evidence of developing of either conditions should regarded as signal of discontinue therapy
Contraindications
Caution in pregnancy.
Caution in lactation.
Caution in Hypersensitivity.
Side Effects
Headache, Flatulence, Dyspepsia, Rash, Allergy, Infection and Back pain.
Storage
Store in the original package in order to protect from moisture.
Pharmacology
Pharmacokinetics
Atorvastatin:After oral administration, Atorvastatin is rapidly absorbed, with peak serum concentrations reaching within 1 to 2 hours. Extent of absorption increases in proportion to Atorvastatin dose. The absolute bioavailability of Atorvastatin is approximately 14% and the systemic availability of HMG-CoA reductase inhibitory activity is approximately 30%. Mean volume of distribution is approximately 381 liters. Atorvastatin is ≥ 98% bound to plasma proteins. Atorvastatin is extensively metabolized to ortho-and para hydroxylated derivatives and various beta-oxidation products. Approximately 70% of circulatory inhibitory activity for HMG-CoA reductase is attributed to active metabolites. Atorvastatin and its metabolites are eliminated primarily in bile following hepatic and/or extrahepatic metabolism; however the drug does not appear to undergo enterohepatic recirculation. Mean plasma elimination half life of Atorvastatin in humans is approximately 14 hrs, but the half life of inhibitory activity for HMG-CoA reductase is 20-30 hours due to contribution of active metabolites. Ezetimibe: Ezetimibe is rapidly absorbed and conjugated after oral administration. Tmax of Ezetimibe and Ezetimibe-glucuronide are 4-12 hrs and 1-2 hrs respectively. Concomitant food administration (high fat or non-fat meals) has no effect on the extent of absorption of Ezetimibe. Ezetimibe can be administered with or without food. Ezetimibe and Ezetimibe-glucuronide are highly bound (>90%) to human plasma proteins. In humans, Ezetimibe gets rapidly metabolized to Ezetimibe-glucuronide. Ezetimibe and Ezetimibe glucuronide are the major drug-derived compounds detected in plasma, constituting approximately 10-20% and 80-90% of the total drug in plasma, respectively. Both Ezetimibe and Ezetimibe-glucuronide are slowly eliminated from plasma with a half-life of approximately 22 hours for both Ezetimibe and Ezetimibe-glucuronide. From the pharmacokinetic profile, it is clear that both the drugs are rapidly absorbed after oral administration. The Tmax for Atorvastatin and Ezetimibe glucuronide (pharmacologically active metabolite of Ezetimibe) are also same (i.e. 1-2 hours) which further supports their use in fixed dose combination as a single dose. Further Atorvastatin and Ezetimibe can be administered as a single dose at any time of the day with or without food.
Interactions
BEFORE TAKING THIS TABLET TELL THE DOCTOR ABOUT ANY MEDICAL PROBLEMS AN ALLERGIES THAT CHILD HAS NOW OR HAS HAD.
- This tablet is the combination of Atorvastatin and Ezetimibe and has unique duel mode of action i.e in this combination, atorvastatin used to treat cardiovascular disease and ezetimibe used to treat high cholesterol. Drug interactions occur when it is taken with another drug or with food. Atorvastatin help to Increases digoxin levels and increases anticoagulant effect of warfarin. Avoid the combination of HMG-CoA reductase inhibitors and fibrates. Ezetimibe: Cholestyramine decreases AUC of ezetimibe by approx 55%, ciclosporin may increase ezetimibe levels.
For Patients
Patient Information
What should I know before taking medicine? Before you take Atenolol Tablets Do not take these tablets if:
- you are allergic to atenolol or to any of the other ingredients of this medicine
- you have low blood pressure
- you have very poor circulation
- you have phaeochromocytoma (high blood pressure caused by a tumour, usually near the kidney) which is not being treated
- you suffer from metabolic acidosis (abnormal levels of acid in your blood)
- you have another heart problem such as heart block or uncontrolled heart failure
- you have a slow or uneven heartbeat
- Before you start taking the tablets, read the manufacturer's printed information leaflet from inside the pack. The manufacturer's leaflet will give you more information about Atenolol and a full list of the side-effects which you may experience from taking it.
- Always take Atenolol Tablets as your doctor has told you.
Chemistry
Atenolol is a selective Β1 receptor antagonist, a drug belonging to the group of beta blockers, a class of drugs used primarily in cardiovascular diseases. Atenolol is used for a number of conditions including: hypertension, angina, long QT syndrome, acute myocardial infarction, supraventricular tachycardia, ventricular tachycardia, and the symptoms of alcohol withdrawal.
Structure
(RS)-2-{4-[2-Hydroxy-3-(propan-2-ylamino)propoxy]phenyl}acetamide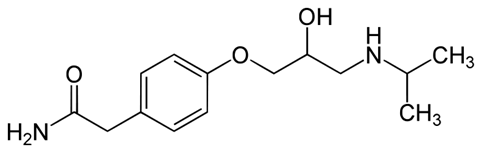
Clinical Data
Chemical DataPregnancy Category
AU: C
US: DLegal status
Rx Prescription only Routes
Oral or IV
Formula
C14H22N2O3
Molecular Mass
266.336 g/mol